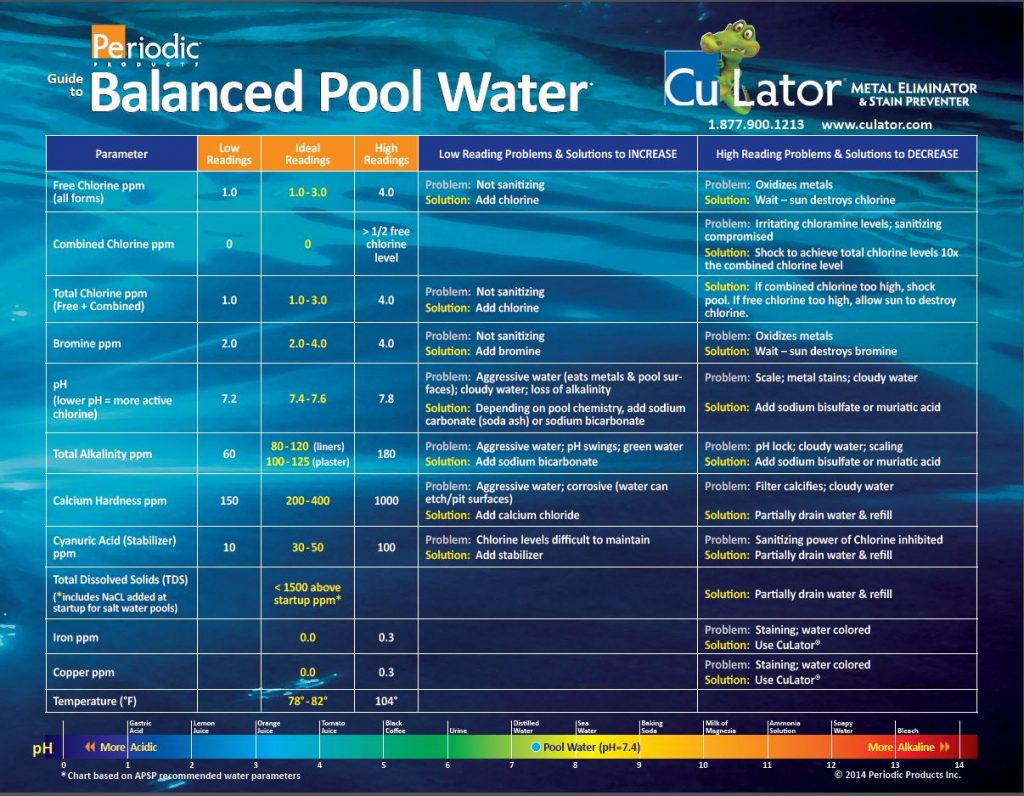
Guide to Balanced Pool Water
Balanced pool water healthy pool water! Follow our easy guide to keep your pool water in perfect balance!
What is balanced pool water?
Balanced pool water is water where all the chemical components added to water are in the optimal balance to create beautiful and safe swimming water.
Why is balanced pool water important?
If one parameter is out of balance, it will affect the other parameters and create problems for the pool and personal safety.
How do I test for balanced pool water?
Have your pool water tested at a local pool store, by your pool professional, or use a pool test kit. You should test at least once a week and re-balance. For better results test every few days and re-balance.
What is the minimum chlorine needed to swim safely?
For residential pools 1.0 free chlorine is needed.
Balanced Pool Water Parameters Explained
*ppm = Part Per Million
Free Chlorine ppm (all forms)
Free “available” chlorine is chlorine in your pool that is ready to kill bacteria and algae. A minimum of 1 ppm is necessary to swim safely in your pool.
Combined Chlorine ppm
Combined chlorine is the chlorine that has already been “used up” sanitizing your water. Chlorine molecules combine with ammonia and nitrogen compounds in the water to form chloramines. Think of chloramines as body waste products combined with chlorine. When your pool smells like chlorine, or your skin smells like chlorine after swimming, you have chloramines and you need to add more free chlorine to sanitize the pool.
Total Chlorine ppm (free chlorine + combined chlorine
If the Total Chlorine in your pool is higher than the Free Chlorine reading, then the difference between the two represents the level of Combined Chlorine. This means the pool is not sanitized properly. If the readings are the same, then no Combined Chlorine is present and your pool is clean. Both Free and Combined chlorine numbers should be the same and above 1 ppm.
Bromine ppm
Sanitizer generally used in spa and hot tubs.
Chlorine Activity is pH Dependent
pH | % Active Form | % Less Active Form |
|---|---|---|
6.0 | 97 | 3 |
6.5 | 91 | 9 |
7.0 | 76 | 24 |
7.2 | 66 | 34 |
7.5 | 50 | 50 |
7.8 | 33 | 67 |
8.0 | 24 | 76 |
8.5 | 9 | 91 |
What is pH?
pH is a measurement of the level of the acidity or basicity of a liquid (water) on a scale of 0-14. pH 7 is neutral. pH Less below 7 is acidic. pH greater than 7 is basic or alkaline. 7.4 to 7.6 ideal for most pool water. pH Influences the ability of chlorine to sanitize pool water.

pH uses a Log scale: 10 x more acid at pH 6 than pH 7 and 100 x more acid at pH 6 than pH 8
pH Problems & Solutions
Low pH (below 7/acidic) causes
- Equipment destruction
- Red eyes
- Hair loss
- Pool surfaces deterioration
- Cloudy water
- Scale removal
- Rapid loss of alkalinity
High pH (above 8/basic) causes
- Scale formation
- Metal staining (plate out)
- Reduced chlorine effectiveness
- Plugged filtration system
- Irritation to users skin and eyes
- Cloudy water
To increase pH add
- Sodium carbonate (soda ash).
- Sodium hypochlorite,
- Calcium hypochlorite,
- Sodium hydroxide (caustic soda),
- Sodium bicarbonate
To decrease pH add
- Sodium bisulfate
- Muriatic acid.
- Acid, trichlor, dichlor, and rain water also lower pH.
Total Alkalinity ppm
Alkalinity is the measure of the pool’s buffering capacity to resist pH change. Water with low alkalinity makes it difficult to maintain pH which unbalance your pool water.
Low alkalinity
- Makes it difficult to maintain desired pH.
- Acts like aggressive water.
- Can be corrosive to equipment and can release metals into the water. (Green water is a symptom of low alkalinity.)
- Is associated with a low pH.
- Can cause eye irritation.
High alkalinity
- Acts like scale-forming water.
- Makes it difficult to adjust pH.
- Can cause pH lock (pH stuck at a certain level and is difficult to change).
- Is associated with a high pH.
- can cause cloudy water
To increase alkalinity
- Add Sodium bicarbonate
To decrease alkalinity
- Add sodium bisulfate
- Add muriatic acid (will also lower pH)
Calcium "Total" Hardness ppm
Low hardness
- Acts like aggressive water.
- Can be corrosive to equipment and can release metals into the water.
- Can etch and pit plaster surfaces, releasing metals into the water.
- Can wrinkle vinyl liners.
High hardness
- Acts like scale-forming water.
- Can cause filter calcification.
- Can cause eye irritation.
- Can cause cloudy water.
To increase hardness
- Add quality calcium chloride.
To decrease hardness
- Drain some pool water. Rain water also reduces hardness.
Total Dissolved Solids (TDS)
TDS is the sum of all of the things dissolved things in a body of water. TDS is everything in the water that is not water. It includes hardness, alkalinity, bromides, cyanuric acid, chlorides, cyanuric acid, sulfates, silicates, and all manner of organic compounds and metals.
Total iron ppm
Iron levels must be performed with a “Total” test to get accurate results. Levels above .3 ppm may cause staining. Ideal level is 0.0.
Total copper ppm
Copper levels must be performed with a “Total” test to get accurate results. Levels above .3 ppm may cause staining. Ideal level is 0.0.
Temperature
As water temperature increases, the water balance becomes more basic and scale-forming. As water temperature decreases, water becomes more corrosive. Temperature should be measured, but cannot be adjusted to achieve saturation or balanced water. Ideal temperature is 78-82 degrees F.